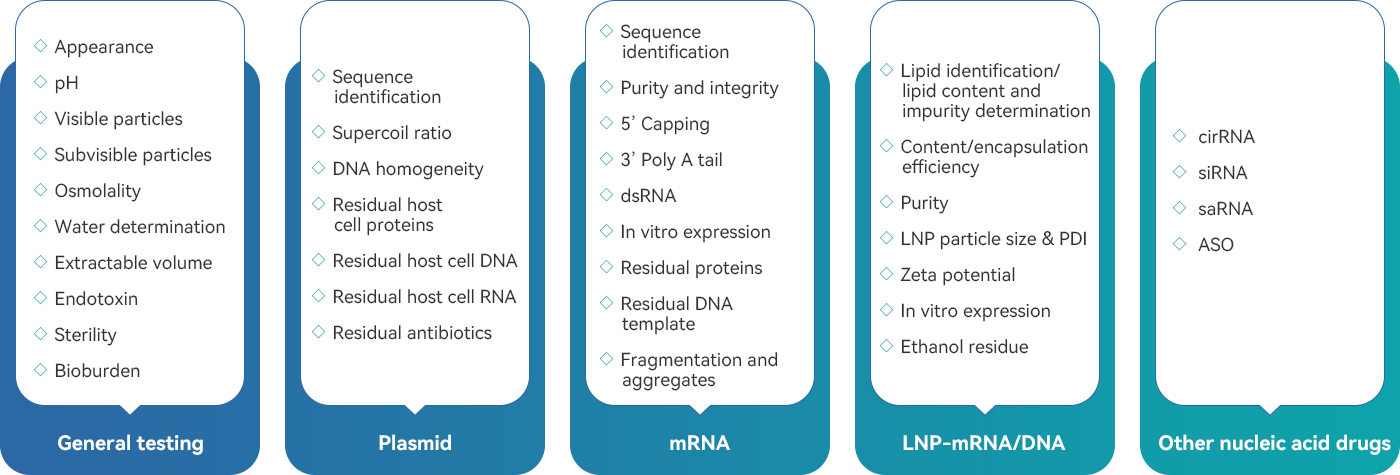
The analytical platform for plasmid and mRNA empowers clients' projects, providing strong analytical support throughout the entire lifecycle of plasmid, mRNA, and LNP-mRNA products, from process development to manufacturing.
The analytical capabilities encompass method development, method transfer, method validation, product quality attribute studies, product release, stability studies, etc. providing strong technical support to accelerate the progression of projects and enabling the regulatory submissions in a short time.
Analytical Methodology
Analytical method development/method transfer/method qualification/method validation
CMC Development Support
Product quality verification, supporting process development and process validation
Product Release/Stability Studies
IPC testing during process development, release testing, long-term/accelerated/stress stability studies
Product Quality Attributes Study
Structural characterization, impurity analysis, in vitro expression, etc.